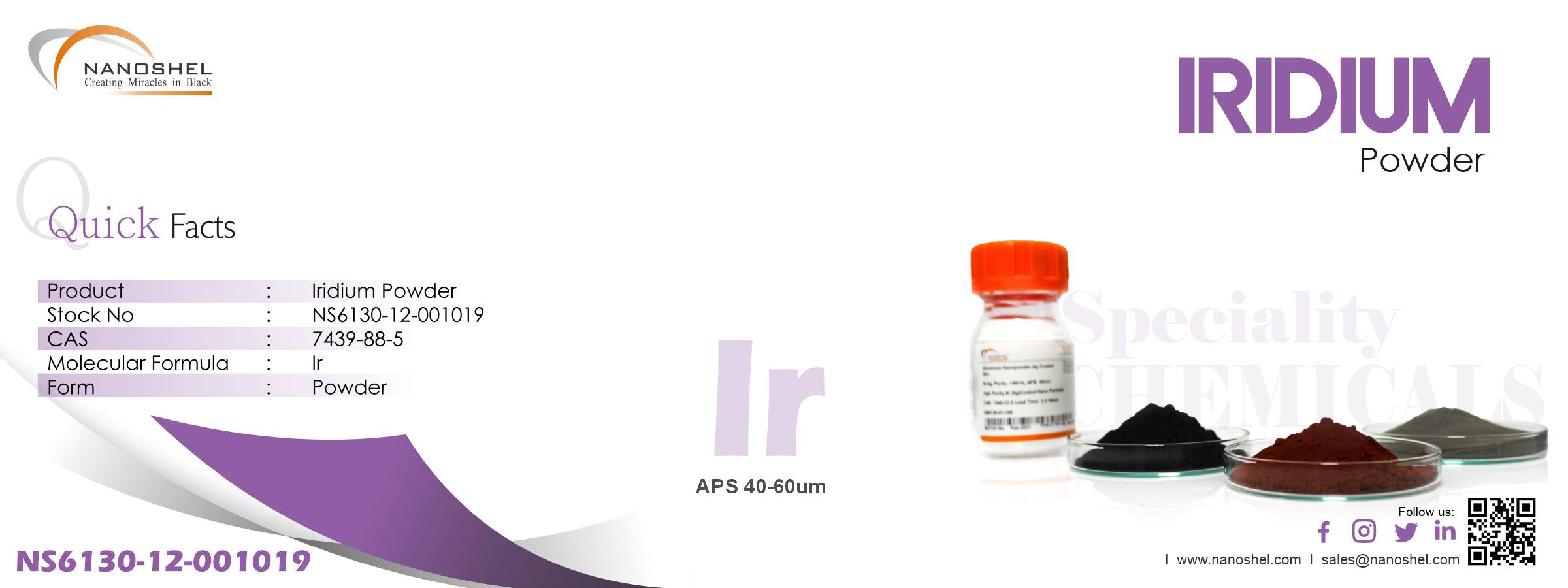
Iridium Metal Powder
Ir, Purity: >99%, APS: 40-60um
Iridium Metal Powder
NS001019
Iridium Metal Powder
| Product | Iridium Powder | |
| Stock No | NS6130-12-001019 | |
| CAS | 7439-88-5 | Confirm |
| APS | 40-60um | Confirm |
| Purity | >99% | Confirm |
| Molecular Formula | Ir | Confirm |
| Molecular Weight | 192.22g/mol | Confirm |
| Form | Powder | Confirm |
| Color | Gray | Confirm |
| Density | 22.56g/cm3 | Confirm |
| Melting Point | 2466°C | Confirm |
| Boiling Point | 4130°C | Confirm |
| Quality Control | Each Lot of was tested successfully | |
| Main Inspect Verifier | Manger QC | |
Typical Chemical Analysis
| Assay | >99% |
| Other Metal | 8000ppm |
Expert Reviews
Iridium Powder
Contact Us
From us, you can easily purchase Iridium Metal Powder - (Ir, Purity: >99%, APS: 40-60um) at great prices. Place an online order and we will dispatch your order through DHL, FedEx, UPS. You can also request a quote by mailing us at sales@nanoshel.com. We invite you to contact us for further information about our company and our capabilities. At Nanoshel, we look forward to your suggestions and feedback.
Email Us:
sales@nanoshel.comcmg@nanoshel.com
🔆
Follow Us:
Direct Call Us:
USA – Sales/Research
+1 646 470 4911
+1 646 470 4911
UK – Sales/Research
+44 1782 454 144
+44 1782 454 144
Ireland – Sales/Research
+354 71 985 3714
+354 71 985 3714
India – Sales/Research
+91-9779550077
+91-9779238252
+91-9779550077
+91-9779238252
Note:
*Exchanges of materials/products are not permitted. Nanoshel does not offer refunds.
*US Dollar Cheques Not Accepted, Only Bank TT/Credit Cards Accepted
*Exchanges of materials/products are not permitted. Nanoshel does not offer refunds.
*US Dollar Cheques Not Accepted, Only Bank TT/Credit Cards Accepted
